Our value-added services include a variety of finish processes, assembly, and packaging. Our goal is to complete your project in the most efficient manner possible.
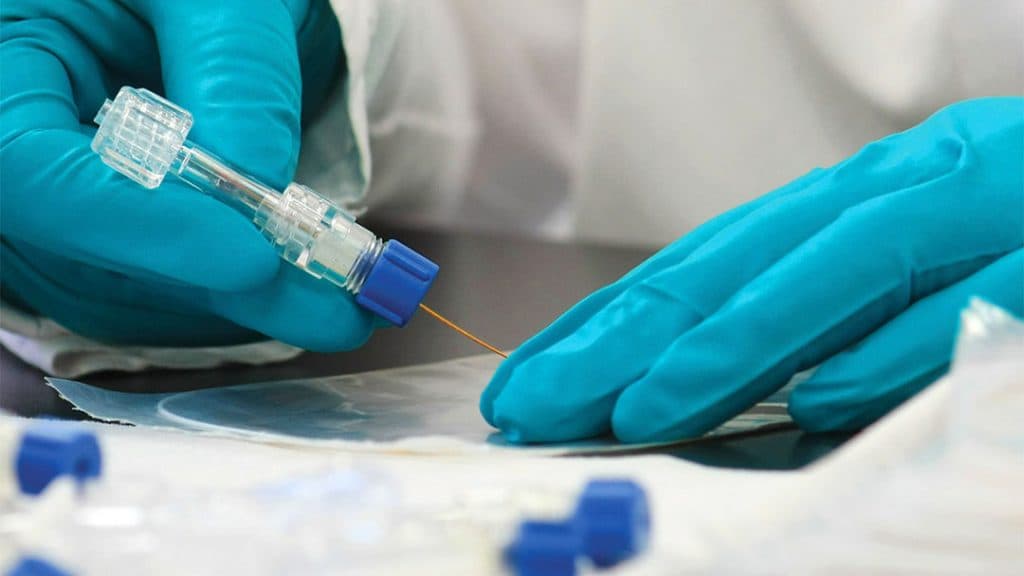
Cleanroom Assembly
CWT’s ISO 13485 Class 7 cleanrooms provide unique and comprehensive manufacturing, assembly and packaging capabilities. Our cleanroom product assembly delivers quality ultra-fine wire parts and sub-assemblies, including custom guidewires, continuous coils and additional medical and dental surgical components as well as other cleanroom-specific components for diverse industries.
Laser & Plasma Welding
We laser weld ultra fine wire components together, with a specialty in small diameter medical wire.
As part of our product assembly and finishing process, we can add specialized laser welding to your project. By incorporating this service into our manufacturing process, we have been able to drastically reduce lead time and supply chain hassles on complex devices.
Our most common welding projects include: stents, hypotubes, guidewires, ball tips, sensors, cannulas and needles, among additional wire devices. Let us know if we can help with your next wire laser weld project.


Laser Marking
Our laser marking capabilities can add unique identifiers to a variety of parts. The identifiers include part numbers, lot numbers, descriptions, and company logos, among many others. Additionally, we can add depth bands to k-wires and pins to give feedback to how far a component may be inserted. We can also mark a variety of materials and sizes. Contact us to learn more about how we can add laser markings to your product.
Ink Pad Printing
CWT offers competitive medical device printing services that are precise, timely, and invaluable. Customers use our pad printing services for a variety of medical product marks. For instance, our medical class black ink can be used for depth marking orthopedic pins or for issuing product number identifiers to accommodate FDA tracking needs. Typical products we print on include catheters, tubing, K-Wires and a variety of stainless steel and nitinol components.


Custom Guidewire Assemblies
As a leading manufacturer of medical guide wires, we can support your required specifications or the contractual needs of OEM products. Production runs are available for prototypes to large contract runs and from bulk & non-sterile assembly to single with cleanroom assembly.
Wire EDM Cutting
Wire EDM Cutting is used to build many of our tooling and fixtures. We have the ability to engineer and draw them in-house using 3D CAD models, and turn that model into a unique file and program that can then be cut use Wire EDM technology. Difficult geometries and materials, such as nitinol, work well with wire EDM cutting. Contact us to learn how we can make your product better using Wire EDM technology.


Passivation
Passivation is the use of a light coat such as metal oxide which is used to create a shell to protect the wire against corrosion. This process removes any free-iron from the surface of stainless steel while creating the passive chromium oxide layer.
We provide passivation for guidewires, k-wires, core wires, coils, mandrels, steinmann pins, and stylets.
Electropolishing
Electropolishing is an electrochemical process which brightens a steel surface by selective dissolution of the high points. Metal is removed from the surface producing a unidirectional pattern that is both stress and occlusion free.
This process can be done for guidewires, k-wires, core wires, coils, mandrels, steinmann pins, and stylets.


PTFE Coating
Using our strategic suppliers we can add PTFE coating to almost any of the products we manufacture. We can apply coatings via spray or dip technology, with a variety of colors and formulations to choose from. Generally, the thickness of our PTFE coatings is ±0.0004±0.0003”. Contact us to learn more about how we can add PTFE coating to your product.



